Secondary nanoparticles formation and composition from open and residential wood burning
Biomass burning is one of the largest combustion-related sources of volatile organic compounds (VOCs) and nanoparticles to the atmosphere. However, the contribution of biomass burning to the regional and global secondary organic aerosol (SOA) burden is quite uncertain, limiting our understanding of its impacts on air quality, climate, and human health. One important issue related to the contribution estimating is the large uncertainty and variation of SOA yield and emission rate, especially the scarce data over long-timescale oxidation. Another issue is the lack of molecular composition and markers which can be used as references for ambient source apportionment. In this work, we studied the long-timescale oxidation of emissions from open and stove wood burning, representing wildfires and residential wood combustion, respectively. By deploying an extractive electrospray ionization time-of-flight mass spectrometer (EESI-TOF-MS) [1], we also provided the molecular-level chemical composition of the formed SOA. The results of this work can largely improve our understanding of the contribution of biomass burning to the SOA burden. Methods. Pine and spruce wood were burned open or in a residential stove, and the burning emissions were injected into a holding chamber. Photooxidation experiments were conducted using a custom-made oxidation flow reactor (OFR) [2]. The burning emissions were introduced into the OFR from the holding chamber with a small flow. Particle size distribution was measured with a scanning mobility particle sizer (SMPS, TSI), and was used to determine the SOA mass concentration. Bulk composition and oxidation state of particles were determined using a long time-of-flight aerosol mass spectrometer (LTOF-AMS, Aerodyne). The molecular composition of particles was measured with an EESI-TOF-MS [1]. Results. SOA mass concentration measured by SMPS and AMS matches very well. Although EESI data has some variations, the general trend in the SOA concentration is very similar: it firstly increases and then decreases with increasing photochemical age. This indicates that fragmentation reactions might play an important role, which can be proved by the decreasing average carbon number (from 10 to 8) measured by EESI with increasing photochemical age (from 0.3 to 7 days). In addition, it is found that there is no significant difference in SOA production from open or stove burning emissions. The carbon and oxygen distributions of the formed SOA show that the majority of SOA are species with carbon number ≤12 and oxygen number ≤7. The time evolution of these species provides insights into the chemistry in the oxidation of biomass burning emissions.
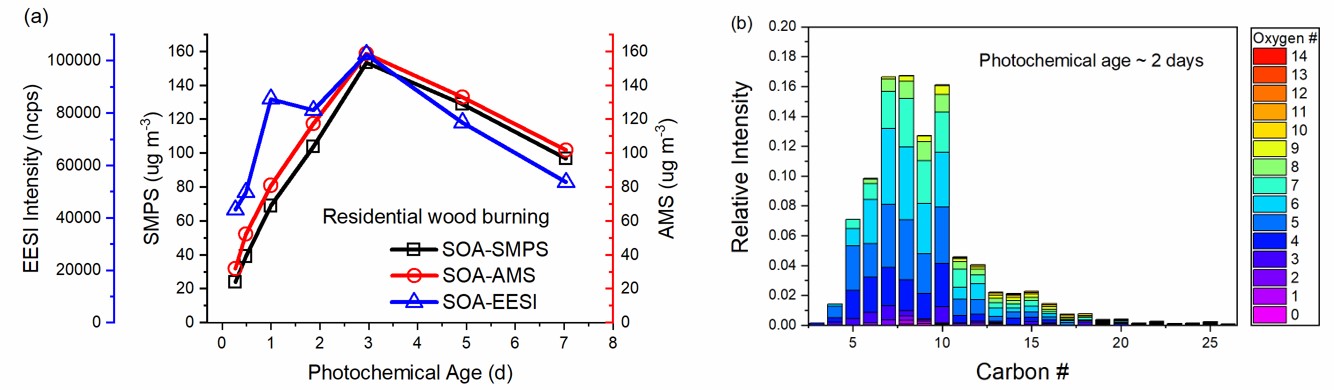
[1] Lopez-Hilfiker, F. D. et al., Atmos. Meas. Tech., 2019, 12, 4867-4886.
[2] Li, K. et al., Atmos. Chem. Phys., 2019, 19, 9715-9731.